Pharmaceutical Container & Packaging Testing
Ensure your pharmaceutical packaging meets the highest safety and regulatory standards. Our comprehensive Pharmaceutical Container & Packaging Testing services deliver critical insights into container integrity, material compatibility, and drug product protection, while our expert problem-solving approach and end-to-end testing capabilities help you navigate complex regulatory requirements and bring safe, compliant medications to market faster.

What is pharmaceutical container and packaging testing at Element?
Pharmaceutical container testing validates the safety and performance of packaging systems used in drug delivery. Container compatibility directly affects medication safety and efficacy, making it a crucial part of product development. Here at Element, we provide comprehensive testing services that ensure your pharmaceutical containers meet rigorous quality and safety standards across multiple container types and materials according to USP, EP, JP, and ISO methods.
What can Element offer you for pharmaceutical container and packaging testing?

Components and materials we test
Components and materials we test
We provide extensive testing across pharmaceutical packaging materials including glass, multiple plastic polymers, PET containers, and various closure systems. Our comprehensive approach covers containers for oral, parenteral, and ophthalmic preparations. Our specialized testing expertise helps ensure that your packaging does not compromise the therapeutic efficacy of your products.
Key tests offered
Key tests offered
- Hydrolytic Resistance
- Material Compatibility Analysis
- Chemical Migration Evaluation
- Performance Integrity Testing
- Environmental Condition Assessments
Element provides full monograph testing services, and we specifically test for:
USP <660> Containers – Glass Type I, II and III Containers:
- Hydrolytic Resistance
- Glass Grains Test
- Surface Glass Test
- Surface Etching Test
USP <661.1> and <661.2> Containers – Plastics HDPE/LDPE/Polypropylene Containers:
- Infrared Spectroscopy (with USP Reference)
- Differential Scanning Calorimetry (with USP Reference)
- Heavy Metals and Nonvolatile Residue
- Buffering Capacity (components used in contact with oral liquids)
Polyethylene Terephthalate (PET) and Polyethylene Terephthalate G (PETG) Containers:
- Infrared Spectroscopy (with USP Reference)
- Differential Scanning Calorimetry (with USP Reference)
- Colorant Extraction
- Heavy Metals
- Total Terephthaloyl Moieties
- Ethylene Glycol
USP <671> - Performance Testing Moisture Permeation Tests:
- Multiple – Unit Containers for Capsules and Tablets (torque only)
- Multiple – Unit Containers for Capsules and Tablets (Without Closures)
- Single – Unit Containers and Unit – Dose Containers for Liquids
- Light Transmission
- Chambers available at 25 °C/40% RH. 23 °C/75% RH
Investigative Failure Analysis, Surface Characteristics
- <1181> Scanning Electron Microscopy / EDS
- <776> Optical Microscopy
EP General Chapters - Plastics:
- (3.1.3) Polyolefines
- (3.1.4) Polyethylene without Additives for Containers for Parenteral Preparations and Ophthalmic Preparations
- (3.1.5) Polyethylene with Additives for Containers for Parenteral Preparations and Ophthalmic Preparations
- (3.1.6) Polypropylene for Containers and Closures for Preparations for Parenteral and Ophthalmic use
- (3.1.15) Polyethylene Terephthalate for Containers for Preparations not for Parenteral use
- (3.2.2.1) Plastic Containers for Aqueous Solutions for Infusion
Container Closures Elastomeric Closures/Rubber Closures:
- (3.1.9) Silicone Elastomer for Closures and tubing
- (3.2.9) Rubber Closures for Containers for Aqueous Parenteral Preparations, for Powders and Freeze-dried powders
Methods and solutions offered
Methods and solutions offered
Element provides pharmaceutical container and packaging testing services to various pharmacopeia methods including USP, EP, and JP methods. Our comprehensive testing methodologies address safety, regulatory compliance, and performance across multiple international pharmacopeia standards. Our engaged experts regularly assist manufacturers encountering packaging-related manufacturing issues, providing proactive failure analysis, as well as expert problem-solving support.
Cutting-edge equipment
Cutting-edge equipment
- Fully-validated autoclave
- Atomic Absorption Spectrometry
- Scanning Electron Microscopy
- Advanced thermal and mechanical testing capabilities
Which labs offer this service
Which labs offer this service
Our team operates from Life Sciences hubs across the world, providing global access to our expert capabilities. Find your nearest Life Sciences hub on our Locations Page.
Standards we test to and products we test
- Glass Containers (Type I, II, III)
- Plastic Containers (HDPE, LDPE, Polypropylene)
- PET and PETG Containers
- Elastomeric Closures
- Rubber Container Closures
Your Challenges, Our Solutions
Drug Safety and Efficacy First
Regulatory Compliance Simplified
Preventing Packaging Failures
Technical Testing Complexity Solved
Why Choose Element

Global Expertise Matters
Comprehensive Single-Source Solution
Proactive Problem-Solving Approach
Technology-Driven Precision
Frequently asked questions
What container types do you test?
We test glass, plastic, PET, and elastomeric containers across multiple pharmacopeia standards.
How do you ensure packaging safety?
Through comprehensive extractables, leachables, and performance testing using advanced analytical techniques.
What is the scope of Element's investigative testing capabilities?
We offer advanced investigative failure analysis, including surface characteristic studies using Scanning Electron Microscopy (SEM), Electron Dispersive Spectroscopy (EDS), and Optical Microscopy techniques to thoroughly examine container and packaging performance.
Do you provide testing for specific pharmaceutical preparation types?
Yes, we offer specialized testing for various preparation types, including containers for parenteral preparations, ophthalmic preparations, aqueous solutions for infusion, oral liquids, capsules, and tablets.
How do you support manufacturers with packaging-related issues?
Our engaged experts regularly assist manufacturers encountering packaging-related manufacturing issues, providing troubleshooting, failure analysis, and proactive problem-solving support to prevent potential packaging failures.
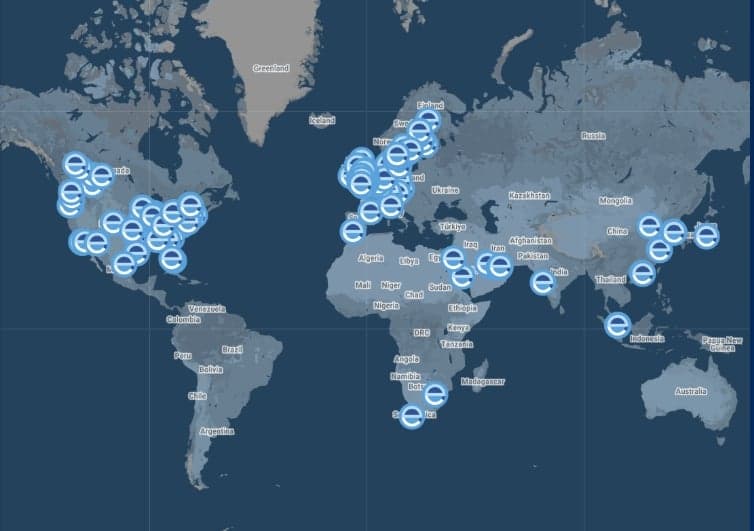
Explore our global network of labs and find your nearest location
VIEW ALL LOCATIONSRelated services

Medical Device Testing Services
With Element as your medical device testing partner, you’ll enjoy the benefit of a single comprehensive supplier across feasibility, R&D and prototype trials, through product development, regulatory validation and production quality control.
Extractables and Leachables Testing Services
Element provides tailored extractables and leachables testing (E&L) studies to ensure patient safety and compliance with regulatory requirements.

Trace Metal Testing and Analysis Services
Element offers trace metal testing and impurity analysis services, addressing safety, compliance, and contamination challenges for pharmaceutical products with advanced techniques and global expertise.

Raw Materials and Excipients Testing Services
Element offers expert raw material and excipient testing services, addressing pharma industry challenges with advanced techniques to support quality, safety, and regulatory compliance throughout drug development and manufacturing.

Material Sciences Testing
Our material testing laboratories provide a complete suite of material testing services for pharmaceutical companies.
