Trace Metal Testing and Analysis Services
Achieve high-quality products and regulatory compliance with precise trace metal analysis. Your products gain reliable safety validation while meeting global standards. Get the accurate data you need for quality assurance, risk assessment, and regulatory documentation.

What Is Trace Metal Testing at Element?
Trace metal testing identifies and quantifies metal elements present at ultra-low concentrations in products and materials. At Element, our analytical techniques detect and measure trace metals with exceptional sensitivity, delivering the data you need for product safety and regulatory compliance.
What can element offer you for Trace Metal Testing and Analysis services

Key Tests Offered
Key Tests Offered
Our comprehensive testing suite covers metals speciation analysis, ICH Q3D and USP 232/233 elemental impurities testing, and unknown identification. We handle specific testing requirements for California Proposition 65 metals, including arsenic, beryllium, cadmium, lead, hexavalent chromium, and mercury compounds. Our USP <2232> analysis ensures dietary supplements meet all permitted daily exposure limits.
Our tests include:
- Metals speciation analysis
- ICH Q3D and USP 232/233 elemental impurities testing
- Pharmaceutical unknown identification and impurity testing
- Pharmaceutical contaminant analysis and complaints investigations
- California Proposition 65 metals testing
- USP <2232> testing of elemental contaminants in dietary supplements and nutraceuticals
Materials and products we test
Materials and products we test
From raw materials to finished products, we analyze pharmaceuticals, biologics, drug substances, medical devices, nutraceuticals, and supplements. Our expertise extends to complex matrices including plastics, lubricants, and specialized pharmaceutical components. We test materials from every stage of manufacturing, including catalysts, processing aids, inorganic materials, container closures, and water sources.
Methods And Solutions Offered
Methods And Solutions Offered
We employ multiple sample preparation techniques optimized for your specific materials:
- Closed-vessel microwave digestion for complete dissolution
- Open-vessel block digestion for complex matrices
- Solvent extraction for organic compounds
- Reflux extraction for difficult materials
- Acid, base, and wet chemical digestion for specialized needs Each method is validated to handle challenging active pharmaceutical ingredients and complex sample matrices.
Metals Speciation Analysis
Element measures metal species at ultra-trace levels using advanced ICP-MS techniques and chromatography methods, enabling detailed risk assessments of elements in drug products. This is critical because different forms of the same element can have vastly different toxicity profiles. This service supports a wide range of industries, ensuring product safety by analyzing both free and bound metal states.
ICH Q3D and USP 232/233 Elemental Impurities Testing
Element provides elemental impurities testing for drug products as required by the FDA, including method development and validation for USP <232>/<233> and ICH Q3D. This ensures compliance with FDA regulations for elemental impurities in pharmaceuticals.
Pharmaceutical Unknown Identification and Impurity Testing
Element identifies and quantifies impurities in drug products, helping assess their safety and efficacy. Our experts analyze organic, inorganic, and residual solvents, ensuring compliance with regulatory exposure limits for both small and large molecules.
Pharmaceutical Contaminant Analysis and Complaints Investigations
Element helps detect and resolve contamination issues in pharmaceuticals by identifying organic and inorganic impurities, solvents, and the root causes of contamination. Our comprehensive services support process improvements and regulatory compliance.
California Proposition 65 Metals Testing
Element provides testing for over 900 chemicals regulated under California's Prop 65 law, including carcinogenic and reproductive toxic elements like arsenic, cadmium, and lead, using ICP-MS for trace elemental analysis.
USP <2232> Testing of Elemental Contaminants in Dietary Supplements and Nutraceuticals
Element offers testing to ensure dietary supplements and nutraceuticals meet USP <2232> guidelines for elemental contaminants. We specialize in testing arsenic, cadmium, lead, and mercury to ensure compliance with permitted daily exposure limits.
Cutting-Edge Equipment We Use
Cutting-Edge Equipment We Use
Our advanced instrumentation includes ICP-MS systems with three collision gases specifically designed to overcome polyatomic interferences, particularly with sulfur and silicon analysis. Our ICP-OES technology delivers exceptional precision, while hyphenated systems couple ICP-MS with LC, GC, and ion chromatography for comprehensive analysis capabilities.
Which Labs Offer This Service
Which Labs Offer This Service
Our team operates from Life Sciences hubs across the world, providing global access to our expert capabilities. Find your nearest Life Sciences hub on our Locations Page.
Standards we test to and materials we test
- USP <232> and <233>
- ICH Q3D
- EP/Ph. Eur. (2.4.20)
- BP and JP methods
- California Proposition 65
- USP <2232>
- Active pharmaceutical ingredients (APIs)
- Drug products and biologics
- Medical devices
- Raw materials and excipients
- Finished products
- Nutraceuticals
- Dietary supplements
- Plastics
- Lubricants
Your Challenges, Our Solutions
From complex regulations to clear compliance
Challenging matrices to reliable results
Ultra-sensitive detection achieved
Quality control mastered
Element Experts at your service
Why Choose Element

Advanced technology leadership
Comprehensive solutions
Ultra-sensitive detection
Quality assurance excellence
Specialized metal speciation analysis
Our metal speciation capabilities evaluate both free elements and bound states, essential for understanding toxicity profiles and material interactions. This specialized analysis is crucial because different forms of the same element can have very different toxicity profiles - for example, hexavalent chromium is much more toxic than trivalent chromium. This supports applications across pharmaceuticals, biologics, food products, and consumer goods through advanced hyphenated techniques combining chromatography with ICP-MS.

Explore our global network of labs and find your nearest location
VIEW ALL LOCATIONSRelated services

Metals Speciation Analysis
Element offers advanced metal speciation analysis, providing detailed data on toxicity and bioavailability to support risk assessments in pharmaceuticals, biologics, and other industries.
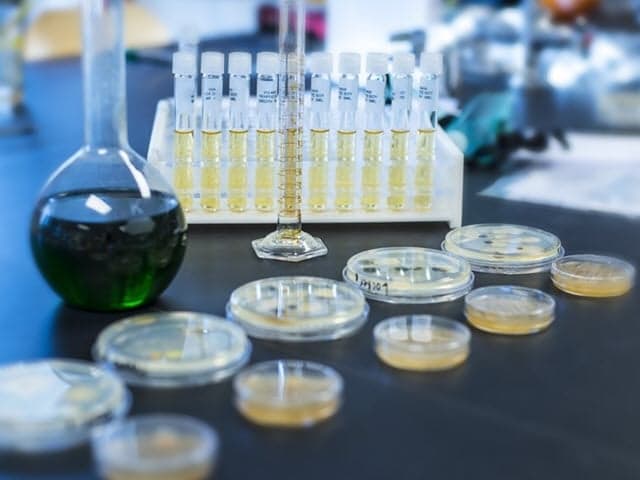
Pharmaceutical Unknown Identification and Impurity Testing
Element offers expert impurity testing services for pharmaceuticals, ensuring drug safety, quality, and compliance with FDA and ICH guidelines through advanced analytical methods and tailored solutions.

Elemental Impurities Testing and Analysis
Industry-leading elemental impurities testing for pharmaceuticals. Offering advanced methods, accurate trace detection, and regulatory expertise to support safe and compliant drug development processes.

