Testing Fracture Toughness of PMMA Bone Cement
Bone cement fixates joints during arthroplasty surgery on knees, hips, shoulders, and elbows by filling the space between the implant and the bone. In this article, we discuss the methodology for measuring fracture toughness of polymethyl methacrylate (PMMA) bone cement.
PMMA is the most common form of bone cement and can come in several different varieties. The chemical formulations can be adjusted, or the cement can be impregnated with antibiotic agents, which can alter the mechanical properties. The most important bone cement functions are to transfer the load from the implant to the bone and absorb shock between the two. As such, understanding the failure modes and the resistance to fracture is critical in evaluating performance.
Bone cements are usually provided as a two-part epoxy, consisting of a powder and a liquid that are mixed. Upon mixing, the bone cement transforms from a runny liquid into a dough-like material that can be molded into the space. An exothermic reaction takes place during the polymerization process, and cures into a hardened cement, fixing the implant in place.
There are several test methods that can be used to evaluate the bone cement properties:
- ASTM F451 - Standard Specification for Acrylic Bone Cement
- ASTM F2118 - Test method for constant amplitude of force controlled fatigue testing of acrylic bone cements materials.
- ISO 5833 - Implants for surgery-Acrylic resin cements
However, this article will specifically outline plane-strain fracture toughness testing of PMMA bone cement using ASTM E399 and ASTM D5045 methodologies.
Evaluating fracture toughness of PMMA bone cement
The purpose of ASTM E399 and ASTM D5045 standards is to determine the fracture toughness (K1C) of metallic or plastic materials. Fracture toughness testing is performed under predominantly linear-elastic, plane-strain conditions. The typical specimen geometries used are compact tension (CT) and single edge notch bending (SENB).
In the example described here, testing was conducted on medical-grade PMMA bone cement using the equations, specimen design and test methods described in ASTM E399 and ASTM D5045. We used a SENB specimen geometry, and a K1C value was reported.
Specimen design
Selecting the proper specimen size and geometry is important to ensure plane-strain conditions at the crack tip are met. A sharp starter notch is needed to ensure a valid fracture toughness value is obtained.
When selecting suitable specimen geometries, several factors are considered, including the amount of material available, cost of material, the orientation of the crack and the difficulty to produce a test specimen. As most bone cement is used in smaller quantities and different geometries, trying to standardize a specimen is difficult as the common sizes have limited physiological relevance. The bone cement test specimen is created by mixing the liquid and powder at the appropriate ratios using the manufacturer’s recommended technique and pouring into a custom mold made to replicate the SENB geometry. This geometry uses less material and makes it easier to machine the material down to the final specimen size.
Specimen fabrication
The custom mold used for creating a specimen is fabricated with food-grade silicone and Delrin material to provide sufficient stiffness (Figure 1). The mold is slightly over-sized and includes integral knife edges to allow for machining once the test specimen has formed and cured. This allows the removal of irregular surfaces and any distortion that occurred while the cement cured. Additional steps are taken to clean and prepare the specimens for testing.

Figure 1: silicone mold (top and middle) and machined test specimen (bottom)
Test methods ASTM E399 and ASTM D5045
The primary focus of this test is to subject the PMMA bone cement to plane-strain conditions. To test plane-strain conditions, a sharp notch is used to initiate a crack in the desired location of the specimen. The crack can be generated by fatigue load, typically referred to as a fatigue pre-crack (ASTM E399) or a razor blade pressed into the specimen, creating a sharp notch and crack (ASTM D5045).
If the cement has brittle behavior, a fatigue pre-crack works best. An SENB specimen is loaded into a 3 point bend fixture (Figure 2), outlined in the ASTM E399 specification, to generate the fatigue pre-crack and conduct the fracture toughness test.

Figure 2: test setup on 3-point bend fixture
If the cement exhibits ductile behavior, the preferred method is that described in the ASTM D5045 specification. This method calls for a razor blade to be pressed into the notch to create a crack similar to the fatigue pre-crack method. It is critical that the notch is straight and extend no less than 0.050” into the specimen when using either the fatigue pre-crack or razor blade method. After a crack has formed penetrating dyes assist in differentiating the crack and the fracture toughness test on the specimen’s fracture surface.
The fracture toughness test uses the same fixture as the fatigue pre-crack method. For PMMA bone cement, the loading rate called out in ASTM E399 is too fast and will cause inconsistent results. A modified displacement rate described in ASTM D5045 is suitable for the material and provided more consistent results. Performing three or more tests for each material condition is recommended.
After specimens are loaded, a load versus loading point displacement curve is generated. The data output is routed through multiple calculations that determine the K1C value.
Reporting and analysis for ASTM E399 and D5045 in brittle materials
ASTM E399 and D5045 are the ideal test methodologies for brittle materials. Three force-displacement modes exist (Figure 3) with a 95% deviation (or secant) from the initial linear portion. On this figure, the point where the 95% deviation crosses the test data is important for determining test validity (it qualifies as a K1C – qualifying that the specimen size was sufficient to plane-strain conditions). As the difference between this 95% deviation intersection and the maximum force increases, the greater the likelihood that the test will be invalid. ASTM E399 allows using an alternative test method if this difference is too large.
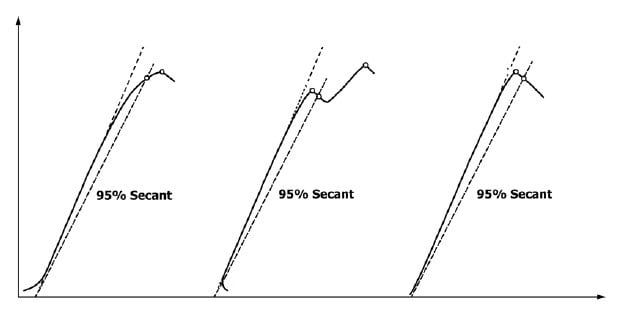
Figure 3: example of three force-displacement modes
Additional factors that influence test validity are pre-crack length, pre-crack straightness, and loading rate. If any of these are invalid, then the final fracture toughness value must be reported as invalid. However, an invalid fracture toughness value does not mean that the data is unusable. The invalid value can be used in the same applications as a valid fracture toughness value, but if a valid fracture toughness value is needed then additional testing is needed to investigate the factors that caused the invalidity. Making changes to testing parameters or specimen size will not guarantee a valid fracture toughness value.
The overall test output (K1C) measures the materials ability to resist fracture in the presence of a flaw which is crucial for understanding the properties of bone cement.
The Element advantage
Our Engaged Experts excel at medical device testing and have worked with many challenging device designs and test setups over the last several decades. Contact us to discuss how we can help with your test project.
Element has one of the most expansive medical device testing scopes in the world, ranging from orthopedics and cardiovascular implants testing to EMC/EMI/product safety testing, and biological and packaging evaluations. We strive to meet all your medical device testing needs in the most expedient, efficient and responsive way.
Related Services

Orthopedic Device Testing Services
Accelerate orthopedic device development with Element's comprehensive testing services. From hip to spinal implants, we ensure regulatory compliance while reducing time-to-market. Expert support at every stage—prototype to commercialization.

Medical Device Testing Services
With Element as your medical device testing partner, you’ll enjoy the benefit of a single comprehensive supplier across feasibility, R&D and prototype trials, through product development, regulatory validation and production quality control.


