What Does Strategic Raw Materials Testing Look Like in Pharmaceutical Manufacturing?
Raw materials testing is the first critical control point in pharmaceutical manufacturing and one of the most consequential. Jihye Jang-Lee, Ph.D., Director of Technical Services at Element, examines the regulatory landscape, the foundations of materials qualification, and how a risk-based approach protects both product quality and operational efficiency.
Critical Decisions at the Supply Chain Gateway
The pharmaceutical supply chain stretches across continents, involving numerous suppliers, manufacturers, and distributors. Without proper verification of incoming materials, manufacturers risk using components that could compromise product quality or patient safety.
Regulatory agencies increasingly scrutinize raw materials testing programs, with expectations extending well beyond acceptance of supplier documentation. The FDA and other regulatory bodies now require comprehensive, risk-based testing programs that demonstrate independent verification of material quality. Recent warning letters highlight heightened regulatory focus on inadequate raw materials testing and over-reliance on supplier Certificates of Analysis (CoA).
Current regulatory expectations include identity testing of every lot, even from qualified suppliers with established track records. For high-risk components such as glycerin and polyethylene glycols (PEGs), verification testing provides essential confirmation beyond supplier documentation. Manufacturers must demonstrate adequate control over elemental impurities, residual solvents, microbial contamination levels, and nitrosamines through appropriate testing protocols.
These requirements reflect regulatory recognition that supplier CoAs, while valuable, cannot substitute for independent verification as part of a robust quality system.
The challenge facing quality professionals becomes clear when considering routine material receipt. A magnesium stearate shipment arrives with supplier documentation showing compliance with all USP specifications. The CoA data appears complete and within range. Quality assurance must design a testing approach that satisfies regulatory requirements, verifies material quality, and supports efficient manufacturing operations.
The most expensive test is the one you didn't perform, until a quality issue emerges downstream.
Raw materials testing serves as the critical first line of defense against contamination, adulteration, or misrepresentation in the pharmaceutical supply chain.
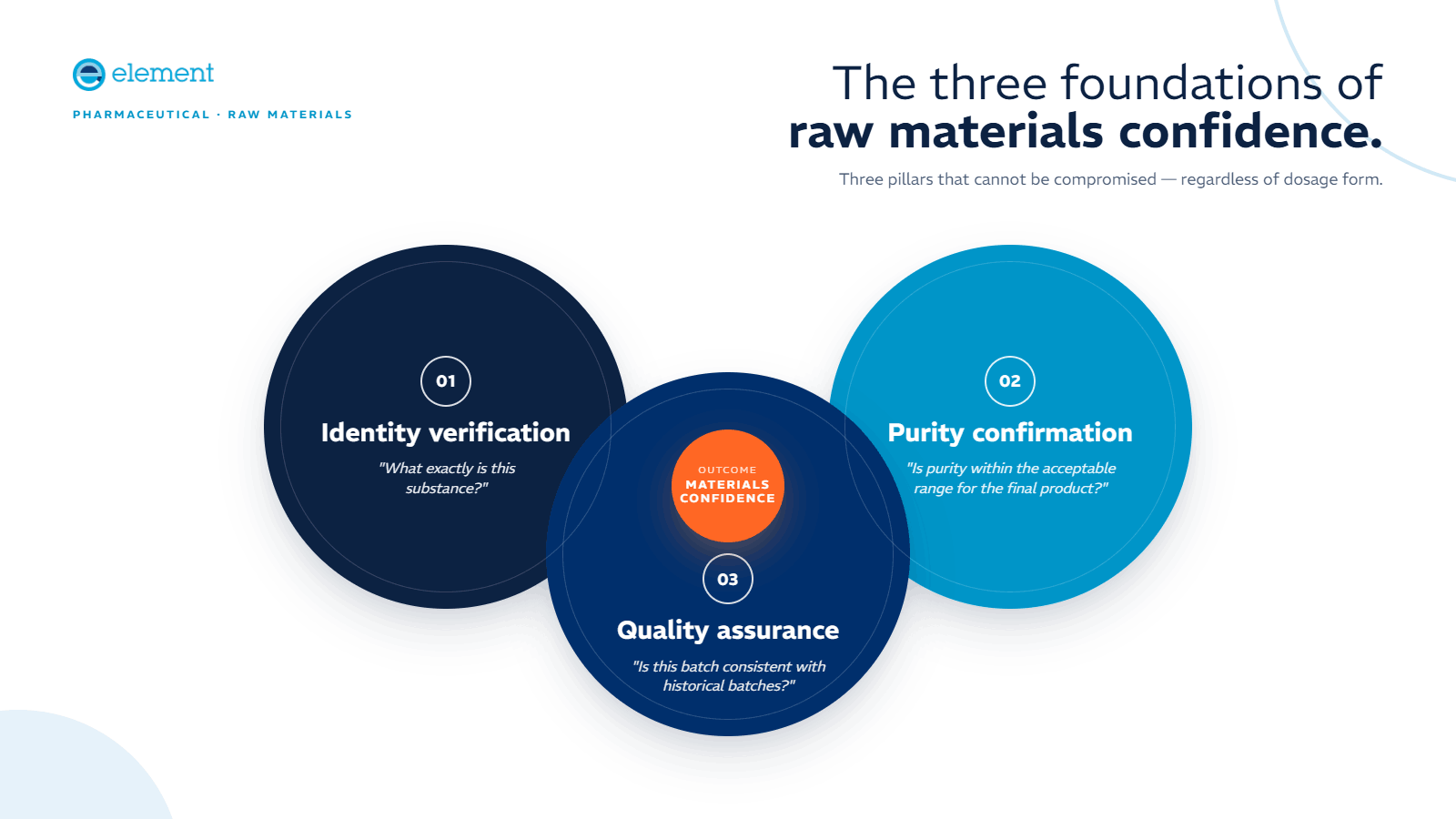
The Three Foundations of Raw Materials Confidence
Pharmaceutical manufacturing stands on three essential pillars that cannot be compromised:
- Identity Verification: What exactly is this substance?
- Purity Confirmation: Is the purity within the acceptable range for manufacturing the final product?
- Quality Assurance: Is this batch consistent with historical batches used in this product?
These factors must be thoroughly understood regardless of dosage form: tablet, syrup, inhaler, or injectable solution. Identity testing isn't optional, even when suppliers are trusted. Materials have been known to travel worldwide under false names, with potentially catastrophic consequences.
Navigating the Quality-Speed-Cost Triangle
The fundamental tension between quality assurance and operational efficiency creates significant challenges for pharmaceutical manufacturers. Quality remains non-negotiable, yet the pressures of production schedules and cost control cannot be ignored.
This tension creates a cascade of interrelated problems:
- Production bottlenecks when testing results aren't available in time to support manufacturing schedules
- Quality blind spots when testing isn't comprehensive enough to catch problematic materials
- Regulatory obstacles when testing methods don't align with constantly evolving requirements
- Market access delays when documentation doesn't satisfy increasingly demanding regulatory reviewers
Compounding these challenges, many facilities lack the specialized equipment and expertise needed for advanced testing methods like USP <232>/<233> trace metal analysis or unknown impurity identification using mass spectrometry. Even well-equipped laboratories struggle to keep pace with evolving regulatory expectations in areas like packaging leachables and data integrity.
When faced with the triangle of quality, speed, and cost, most companies choose two. The best find ways to optimize all three through strategic testing approaches.
From Compliance Checkboxes to Strategic Material Intelligence
Effective raw materials testing isn't about running a standard battery of tests on every incoming material. It requires a thoughtful, risk-based, systematic approach that addresses the specific risks of each material based on criticality, supplier history, and intended use.
The testing process moves through progressive stages:
- Identity verification: established techniques like infrared spectroscopy (IR), UV analysis, and specific optical rotation tests confirm the nature of the material.
- Potency and purity analysis: chromatographic techniques (HPLC for non-volatile compounds, GC for volatile and semi-volatile substances) determine strength and quantify impurities.
- Physical properties: appearance, pH, volatility, melting point, and optical characteristics provide initial insights into material consistency.
- Contaminant analysis: known and unknown impurities, particulates, residual solvents, and trace metals, perhaps the most technically challenging aspect.
A strategic approach asks not just "What tests are required?" but "What information do we need to protect our patients and our products?"
Building a Pharmaceutical Raw Materials Testing Program That Works for Your Supply Chain
Understanding what strategic raw materials testing requires is the foundation. Bringing it to life, the analytical methods, quality systems, and practical implementation decisions are covered in the companion article in this series, with full detail in the accompanying whitepaper.
For a comprehensive exploration of the full topic, download the white paper, Ensuring Pharmaceutical Quality Through Strategic Raw Materials Testing, authored by Jihye Jang-Lee, Ph.D.
Element's network of testing laboratories provides access to advanced instrumentation, specialized expertise, and flexible capacity to support pharmaceutical manufacturers at every stage of their raw materials qualification programs. Learn more about Element's pharmaceutical testing capabilities or contact our experts to discuss your needs.
Related Services

Raw Materials and Excipients Testing Services
Element offers expert raw material and excipient testing services, addressing pharma industry challenges with advanced techniques to support quality, safety, and regulatory compliance throughout drug development and manufacturing.

Raw Material, In-Process & Lot Release Testing services
Our raw material and finished product testing bring peace of mind to manufacturers of drug products ensuring the safety, purity, and quality of both starting materials and manufactured products.

Compendial Monograph and Pharmacopeial Testing
Element provides expert compendial testing services to help pharmaceutical and biotech companies meet global pharmacopeial standards, ensuring product quality, compliance, and market readiness.

Pharmaceutical Container & Packaging Testing
Element provides professional testing services for containers, container closures, and pharmaceutical packaging on polymers and glass containers to several pharmacopeia methods.

Pharmaceutical Quality Control Testing
Element provides pharmaceutical quality control testing, covering raw materials, APIs, and finished products. Our expert analytical development and validation help meet regulatory requirements and support all phases of product development.

Chemical Characterization
Element's chemical characterization services help identify, analyze, and ensure the quality of materials, addressing challenges in purity, composition, and contaminants with advanced techniques and expertise.



